www.hik-consulting.pl - Aparatura Pomiarowa, Stanowiska Szkoleniowe
Stała Plancka - Stanowisko Edukacyjne
Stała Plancka (oznaczana przez h) – jedna z podstawowych stałych fizycznych. Ma wymiar działania, pojawia
się w większości równań mechaniki kwantowej. Historycznie stała Plancka pojawiła się w pracy Maxa Plancka na temat wyjaśnienia przyczyn tzw. katastrofy w nadfiolecie w prawie promieniowania ciała doskonale czarnego. Planck stwierdził, że energia nie może być wypromieniowywana w dowolnych ciągłych ilościach, a jedynie w postaci „paczek” (kwantów)
o wartości hν, gdzie ν jest częstotliwością.
się w większości równań mechaniki kwantowej. Historycznie stała Plancka pojawiła się w pracy Maxa Plancka na temat wyjaśnienia przyczyn tzw. katastrofy w nadfiolecie w prawie promieniowania ciała doskonale czarnego. Planck stwierdził, że energia nie może być wypromieniowywana w dowolnych ciągłych ilościach, a jedynie w postaci „paczek” (kwantów)
o wartości hν, gdzie ν jest częstotliwością.

Opis Stanowiska:
Highlighting the photoelectric effect, emission
of electrons by a metal material when the latter is exposed to electromagnetic radiation of a sufficiently high frequency, is a means of approaching and better understanding the particulate aspect of light. The experiment proposed, using our photoelectric cell, a high pressure mercury spectral lamp and a micro-ammeter, aims at showingthe photoelectric effect, verifying Einstein’s equation and experimentally measuring Planck’s constant. To do this, the photoelectric cell, with a spectral range from approx. 340 to 700 nm, is installed in a metal box ensuring its light insulation and safety. A drum secured on the front of this box allows a variety of filters and diaphragms to be placed in front of the cell to perform a set of measurements. Last but not least, one device only is used to supply both the cell anode and cathode with two voltage ranges (-2/2 V and -2/30 V) and as micro- ammeters to measure photo-current (with a number of measurement ranges: 10-8 to 10-13 A).
MEASURING THE CURRENT VOLTAGE CHARACTERISTIC OF THE PHOTOELECTRIC CELL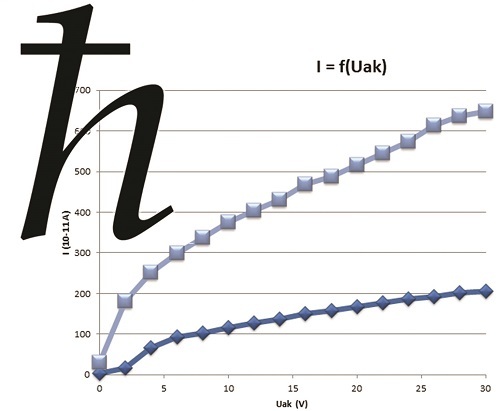

Photo-current value increases at the same time as the value of the difference in electric potential applied between the cathode and the UAK anode. This photo-current reaches saturation when UAK attains a certain value. This level of saturation depends on light intensity, but not on incident frequency. When UAK becomes negative, photo-current drops. Based on this experiment, we can measure a number of photoelectric cell current-voltage characteristics according to light intensity (with diaphragms) and to incident frequency using the mercury lamp (using the filter set). This is a means of checking experimentally matching with the theoretical characteristic.
EXPERIMENTAL DETERMINATION OF THE VALUE OF PLANCK'S CONSTANT h
According to Einstein’s theory, light is composed of a flux of particles, known as photons. The energy of each photon is then equal to E = h.í, where h is Planck’s constant. Measurement of Planck’s constant based on the photoelectric effect relies on determination of the difference in breaking voltage (for which photocurrent is zero) for various homogeneous incident frequencies. To do this, we propose using the “zero current” or “compensation” method. In the 10-13 A measurement range, we modify the potential value until a zero photocurrent is obtained. The spectral lamp is then covered, and current is measured. This current value is very close to dark current and photoelectric cell noise. The lamp is uncovered, and potential adjusted to attain the current value previously measured. We thus obtain breaking potential according to incident frequency. Based on the potential – frequency straight line slope;
the value of h can be calculated experimentally.
SUBJECTS APPROACHED
- Determining h
- Implementing a protocol
- Measuring the current-voltage
characteristic of the photoelectric cell
NECESSARY EQUIPMENT
- POD068951 Planck constant pack
(photo electric cell,
power supply/ ammeter ) x1 - POD010057 Mercury spectral lamp,
high pressure x1 - POD010056 Stand for spectral lamp x1
- POD002192 Half-moon stand x1
Stanowisko dostarczamy wraz z propozycjami ćwiczeń.


